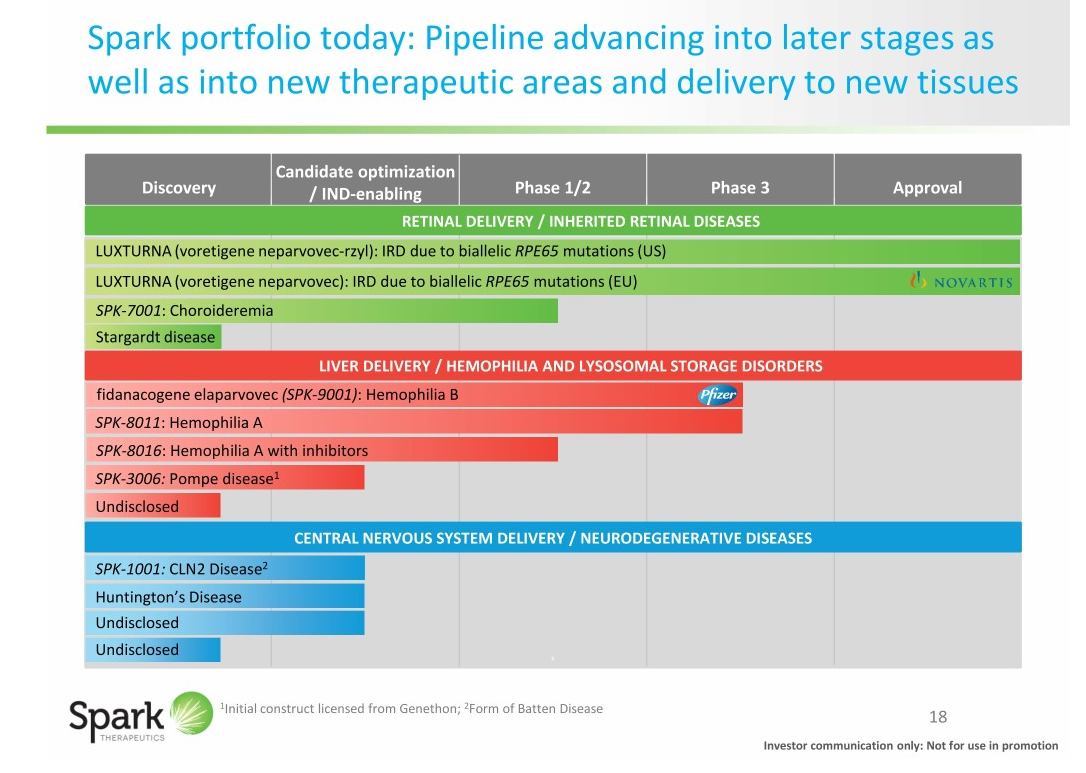
Recommended location of subretinal injection is along the superior vascular arcade, at least 2 mm distal to the center of the fovea to create a subretinal bleb using a 41-gauge subretinal injection cannula with a polyamide micro tip. The Diluent is supplied in two single-use 2-mL vials. The supplied concentration (5x10 12vg/mL) requires a 1:10 dilution prior toĪdministration. recommends a single dose for each eye of 1.5 x 10 11 vector genomes (vg), administered by subretinal injection in a total volume of 0.3 mL. Voretigene neparvovec-rzyl (Luxturna™) is indicated for the treatment of patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy with viable retinal cells. Phase 3 results at 3 and 4 years confirmed these findings with maintenance of overall improvement in ambulatory navigation, light sensitivity, and visual field with no serious adverse events or deleterious immune responses. The study showed evidence of long term safety (4 years) with improved navigational ability (Multi-Luminance Mobility Test (MLMT)) and light sensitivity (full-field light sensitivity threshold (FST) testing). Phase 1 and phase 3 subjects were studied for long term efficacy and safety. Biallelic variations in RPE65 lead to photoreceptor degeneration and can cause severe retinal dystrophies. The RPE65 gene is located on the short (p) arm of chromosome 1 at position 31 ( 1p31.3). 
Retinitis pigmentosa (RP), Leber congenital amaurosis (LCA) and Fundus albipunctatus have subtypes related to pathogenic variants in RPE65. RPE65 (retinal pigment epithelium–specific protein 65-kD) gene encodes an enzyme in the retinal pigment epithelium (RPE) that is responsible for regeneration of 11-cis retinol in the visual cycle. Gene therapy for RPE65 variant-associated retinal dystrophy uses AAV2 vectors to transfect cells with a functioning copy of RPE65 in the RPE cells. The presence of the blood-retinal-barrier can also help decrease the risk of unintentionally spreading vectors to neighboring tissues as well as to the general circulation. The structure and accessibility of the retina, in addition to its immune privileged properties that limit inmune response, make it an ideal target organ for genetic therapies. Recombinant AAV2 is the most popular serotype in AAV-based research and clinical trials. Of the 12 AAV serotypes identified so far, AAV2, AAV4, and AAV5 are specific for retinal tissues. Adeno-associated viruses (AAV) are used in gene therapy due to their unique biology, simple structure and poorly immunogenic response compared with other viruses, but can still trigger an immune response to the virus or the modified protein. Genes may be introduced into cells through a vector, usually a virus. Gene therapy can be defined as the genetic modification of DNA to produce a therapeutic effect by replacing a mutated gene, adding a missing gene or modifying an existing one.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
